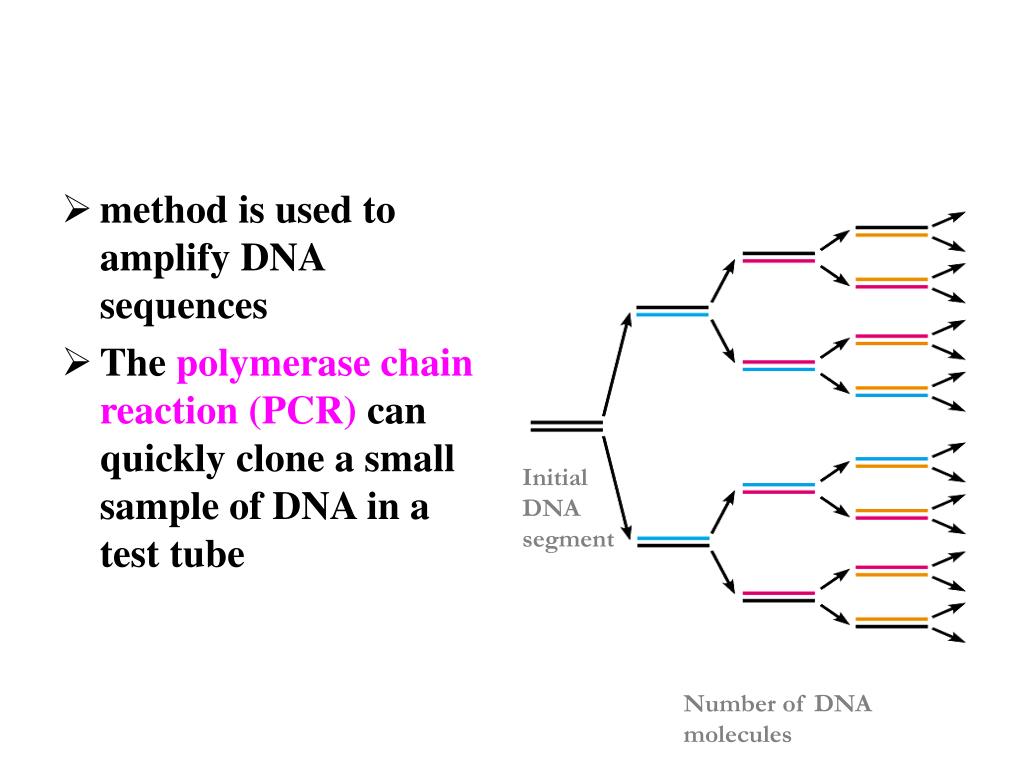
T m 55–70☌ (within 5☌, for two primers).Equally important, the primers should be designed without complementarity between the primers (especially at their 3' ends) that promotes their annealing (i.e., primer-dimers), self-complementarity that can cause self-priming (i.e., secondary structures), or direct repeats that can create imperfect alignment with the target area of the template. First, primer sequences should possess melting temperatures (T m) in the range of 55–70☌, with the T ms of the two primers within 5☌ of each other. In addition to sequence homology, primers must be designed carefully in other ways for specificity of PCR amplification. As such, the primers’ binding sites must be unique to the vicinity of the target with minimal homology to other sequences of the input DNA to ensure specific amplication of the intended target. During PCR, DNA polymerase extends the primers from their 3′ ends. PCR primers are designed to bind (via sequence complementarity) to sequences that flank the region of interest in the template DNA. PCR primers are synthetic DNA oligonucleotides of approximately 15–30 bases. These enzymes are capable of generating lower-error PCR products from long templates in a shorter time with better yields and higher resistance to inhibitors (learn more about DNA polymerase characteristics). However, nonspecific PCR products may appear with higher enzyme concentrations ( Figure 2).įor more specialized applications such as PCR cloning, long amplification, and GC-rich PCR, DNA polymerases with higher performance are preferred. For example, when inhibitors are present in the DNA sample, increasing the amount of DNA polymerase may improve PCR yields. However, it may be necessary to adjust the enzyme amounts with difficult templates. In a typical 50 µL reaction, 1–2 units of DNA polymerase are sufficient for amplification of target DNA. Nowadays, new generations of DNA polymerases have been engineered for greatly improved PCR performance. It incorporates nucleotides at a rate of about 60 bases per second at 70☌ and can amplify lengths of about 5 kb, so it is suitable for standard PCR without special requirements. Taq DNA polymerase has relatively high thermostability, with a half-life of approximately 40 min at 95☌. Taq DNA polymerase is arguably the best-known enzyme used for PCR-its discovery revolutionized PCR. With optimized PCR purification kits, the PCR clean-up procedure can be performed in as little as 5 minutes.ĭNA polymerases are critical players in replicating the target DNA. For best results, PCR amplicons should be purified before re-amplification. To avoid such inhibition, a general recommendation is to dilute the reaction in water prior to the next round of PCR. Although unpurified products may be directly used as a template, carryover reaction components such as primers, dNTPs, salts, and by-products can adversely affect amplification. Also, the selected DNA polymerase should be certified for controlled low level of residual DNA, to minimize false signals in PCR.īesides gDNA, cDNA, and plasmid DNA, it is also possible to re-amplify PCR products to obtain a higher yield of the target. In practice, however, amplification efficiency of a specific template amount is highly dependent upon reaction components and parameters, as well as sensitivity of the DNA polymerase. In theory, a single copy of DNA or a single cell is sufficient for amplification by PCR under ideal conditions. For convenience and simplicity, an online tool is available to calculate copy number from the mass of the input DNA. The molar mass of a particular DNA strand is determined by its size or total number of bases (i.e., a combination of its length and single-stranded or double-stranded nature). Using Avogadro’s constant (L) and molar mass, copy number can be calculated as:Ĭopy number = L x number of moles = L x (total mass/molar mass) The copy number calculation depends on the number of molecules present, in moles of DNA input. At times, PCR protocols may call for input of DNA in terms of copy number, especially for gDNA.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
